|
These data further support the potential of pembro + chemo as a first-line treatment option for metastatic TNBC. The study evaluated pembrolizumab plus investigator’s choice of one. KEYNOTE-355 was an international randomized, placebo-controlled phase III trial conducted at 209 sites in 29 countries. A trend toward improved efficacy with PD-L1 enrichment with pembro + chemo was observed for ORR, DCR and DOR endpoints. Corts presented overall survival and the analysis stratified by levels of PD-L1 expression as assessed by CPS score. Results for the key secondary endpoints of ORR, DCR, and DOR favored pembro + chemo, with the treatment effect increasing as CPS increased ( Table).Ĭonclusion: In subgroup analysis, PFS with pembro + chemo compared to pbo + chemo in pts with metastatic TNBC was improved regardless of chemo partner. The HR for PFS favored pembro regardless of choice of chemo or CPS population ( Table). Results: As of Dec 11 2019, median follow-up was 25.9 mo for pembro + chemo (n=566) and 26.3 mo for pbo + chemo (n=281). The PFS treatment effect was assessed in subgroups descriptively using HRs and 95% CIs although subgroup analysis by on-study chemo were pre-specified, the trial was not powered to compare efficacy among treatment groups by different chemo regimens. Secondary endpoints include ORR, DCR (CR + PR + SD ≥24 weeks), and DOR. Dual primary endpoints are PFS (RECIST v1.1, blinded independent central review) and OS in pts with PD-L1 positive tumors (CPS ≥10 and ≥1) and in all pts. Pts were stratified by chemo type (taxane vs gemcitabine/carboplatin), PD-L1 status (CPS ≥1 vs <1), and prior (neo)adjuvant treatment with same-class chemo (yes vs no). 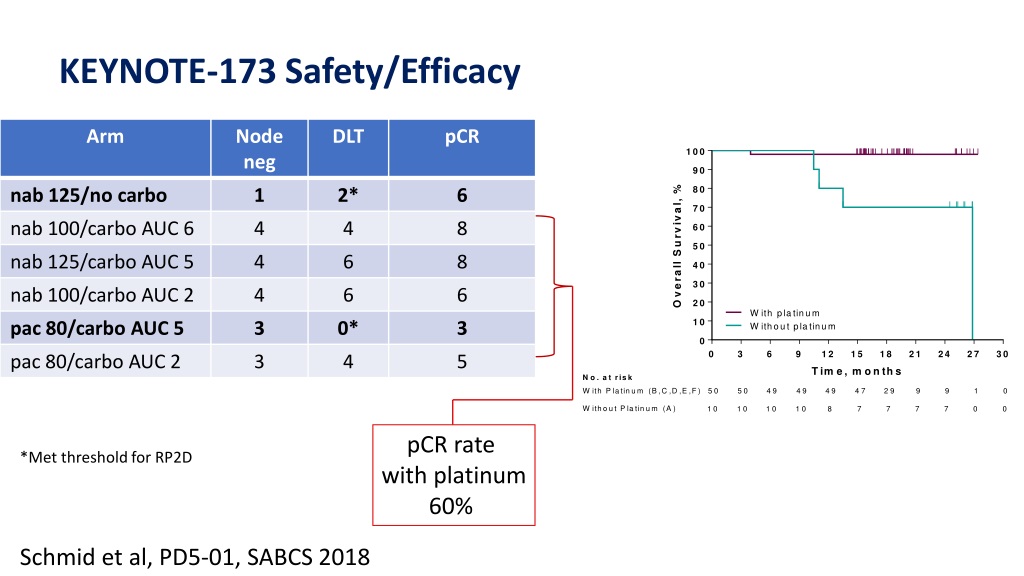 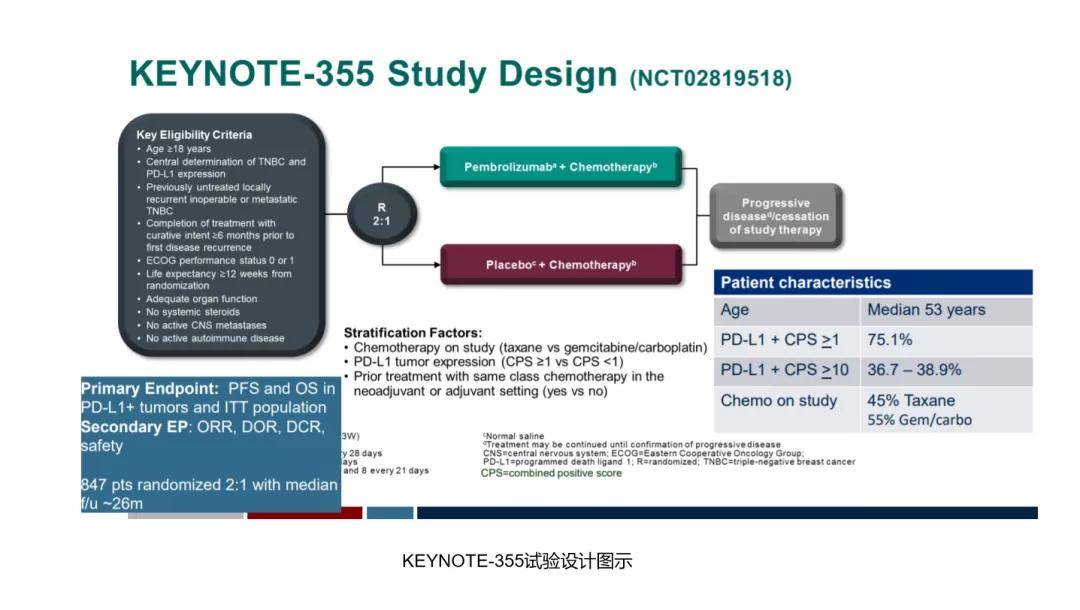
Steroid premedication for paclitaxel was given according to local guidelines and practices, and was not restricted by the protocol. 
Methods: 847 pts with measurable disease per RECIST v1.1, ECOG PS 0-1, and ≥6 mo DFI were randomized 2:1 to pembro + chemo (nab-paclitaxel 100 mg/m 2 days 1, 8, and 15 every 28 days paclitaxel 90 mg/m 2 days 1, 8, and 15 every 28 days or gemcitabine 1000 mg/m 2 + carboplatin AUC 2 days 1 and 8 every 21 days) or pbo + chemo for up to 35 administrations of pembro/pbo or until progression/intolerable toxicity. Here, we examine PFS outcomes for each chemo partner and present key secondary endpoints from KEYNOTE-355. Additionally, pembro + chemo was generally well tolerated, with no new safety signals. In a prespecified interim analysis of KEYNOTE-355 (NCT02819518), pembro combined with chemotherapy (chemo) showed a statistically significant improvement in PFS versus chemo alone in pts with previously untreated locally recurrent inoperable or metastatic TNBC whose tumors expressed PD-L1 CPS ≥10 (HR for progression or death, 0.65, 95% CI, 0.49-0.86 one-sided P=0.0012 ). Background: Pembrolizumab (pembro) monotherapy showed durable antitumor activity and manageable safety in patients (pts) with metastatic triple-negative breast cancer (TNBC) in the KEYNOTE-012, -086, and -119 studies.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
